
the number of nuclear protons, equivalent to the element number), rather than atomic weight, became the recognised basis for ordering its members. 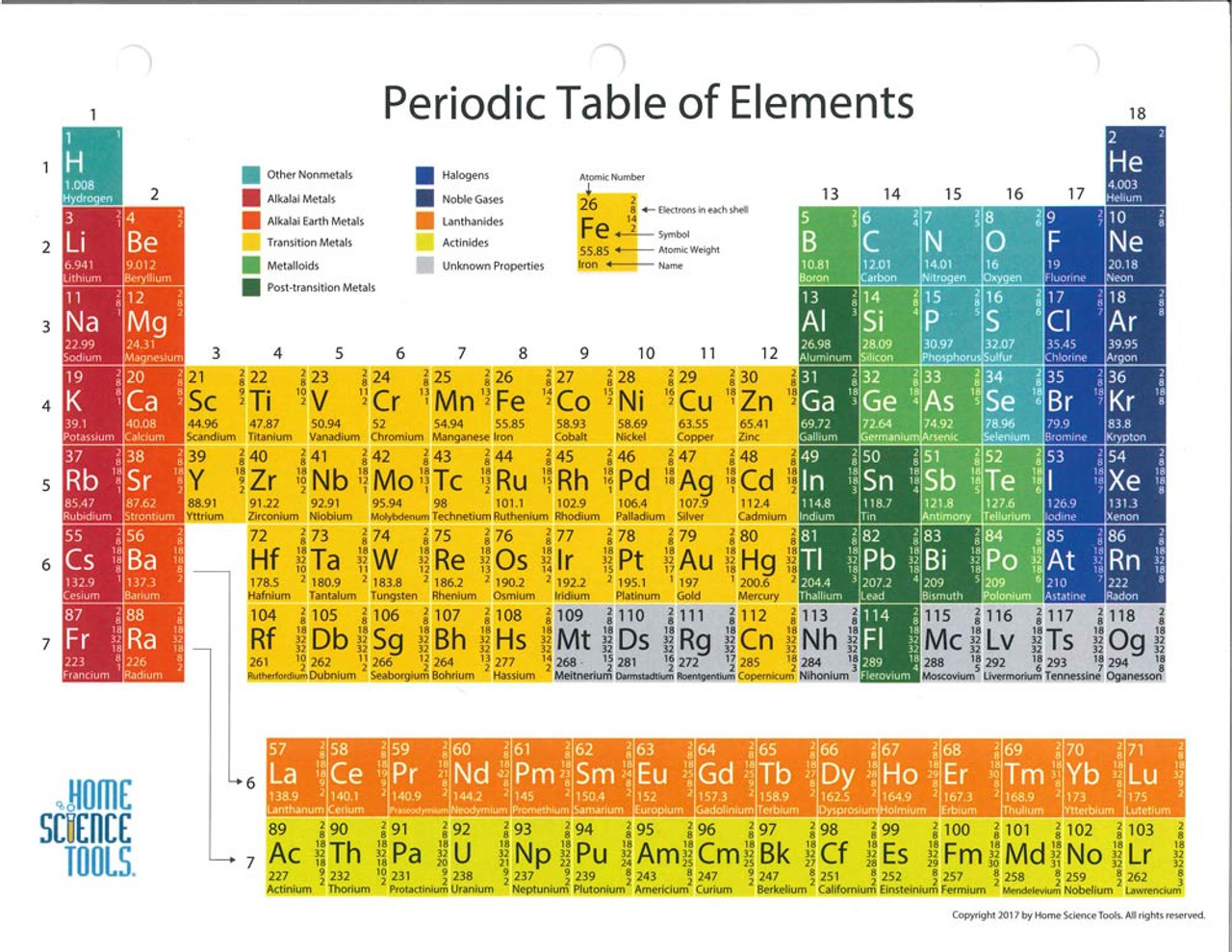
The underlying justification for the structure of the Periodic Table emerged only many years later when atomic structure became understood and atomic number (i.e. From his Periodic Table Mendeleev predicted the properties of then unknown elements such as gallium (element 31), germanium (32), scandium (21) and technetium (43), represented by the gaps, which were later discovered, 4 and subsequently others like Henry Moseley 5 continued to extend the Periodic Table through predictions and by filling the gaps. 3 Mendeleev was not the first to publish listings of the known elements in a table but, building on and surpassing the earlier efforts, he rigorously applied the available knowledge of periodic trends in the relationships between the then approximately 60 known elements to produce a chart in which there were gaps at some points. This was an innovative advance in classification which has helped guide the understanding of chemistry and has spurred on advances in the theoretical understanding of atomic structure. The International Year marks the 150th anniversary of the publication by the Russian chemist Dmitry Mendeleev (1834–1907) of his Periodic Table 2 and celebrates the significance and impact of this outstandingly successful chart of the atomic building blocks of matter. The designation by the UN of 2019 as the International Year of the Periodic Table of Chemical Elements 1 provides a timely opportunity to reflect on this warning and consider how best to meet the challenge it presents. We need to ensure that the finite stocks of these are not excessively depleted or used in environmentally damaging ways. This fact has key implications for the building up of the periodic table of elements.The Table reminds us that there are less than 100 stable elements on our planet (as well as a couple of dozens of radioactive ones) from which to derive all the materials that are required for life and for well-being and comfortable living. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. 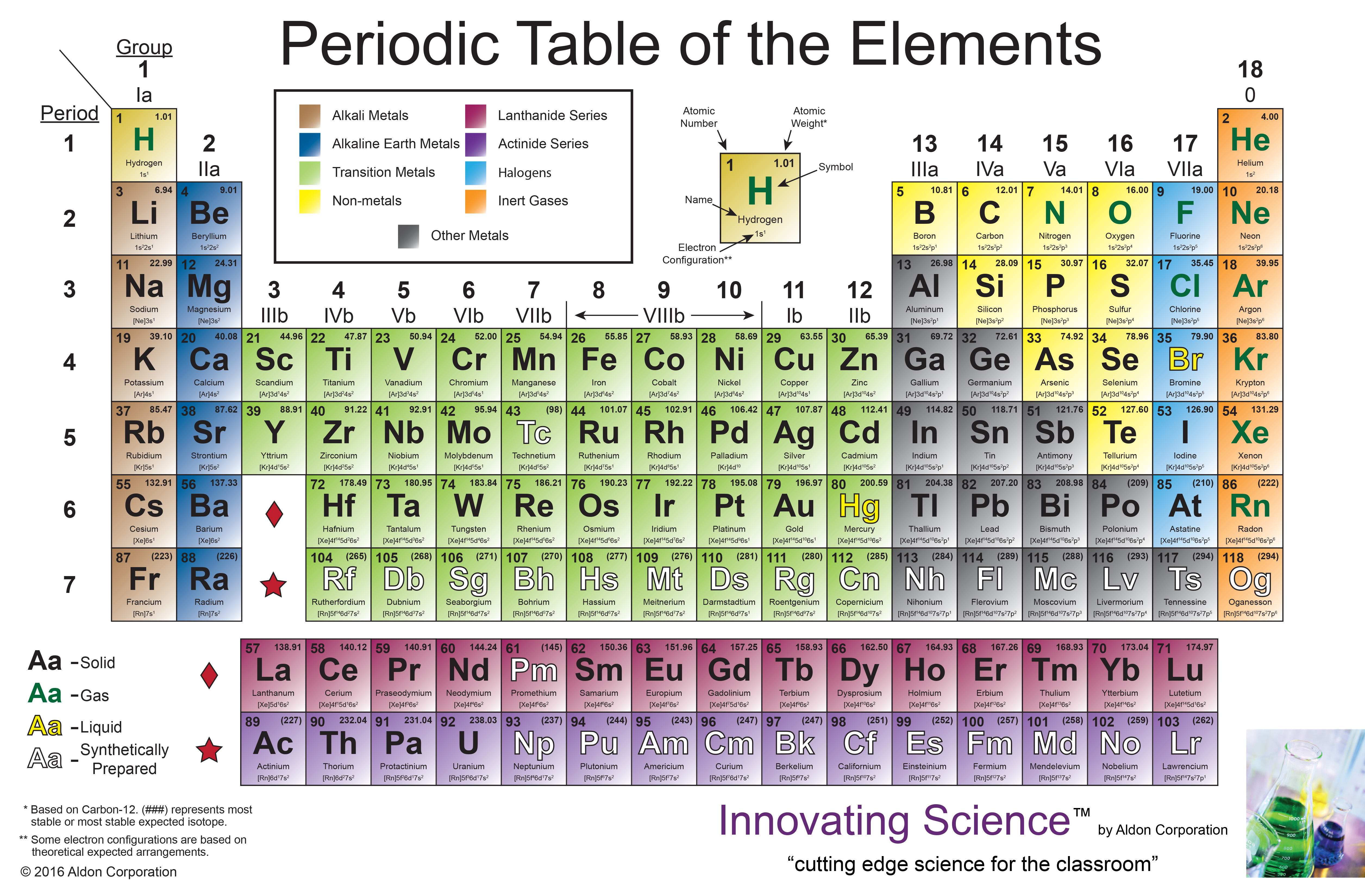
The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol Z. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. There is a recurring pattern called the “periodic law” in their properties, in which elements in the same column (group) have similar properties. Generally, within one row (period) the elements are metals to the left, and non-metals to the right, with the elements having similar chemical behaviours placed in the same column.Įvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is organized in order of increasing atomic number. The periodic table is a tabular arrangement of the chemical elements.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
